13, which showed that, in early stage, untreated non–small-cell lung cancer, selection pressure from antitumor immune responses resulted in multiple routes of tumor immune evasion. Immunoediting was beautifully demonstrated in ref. 
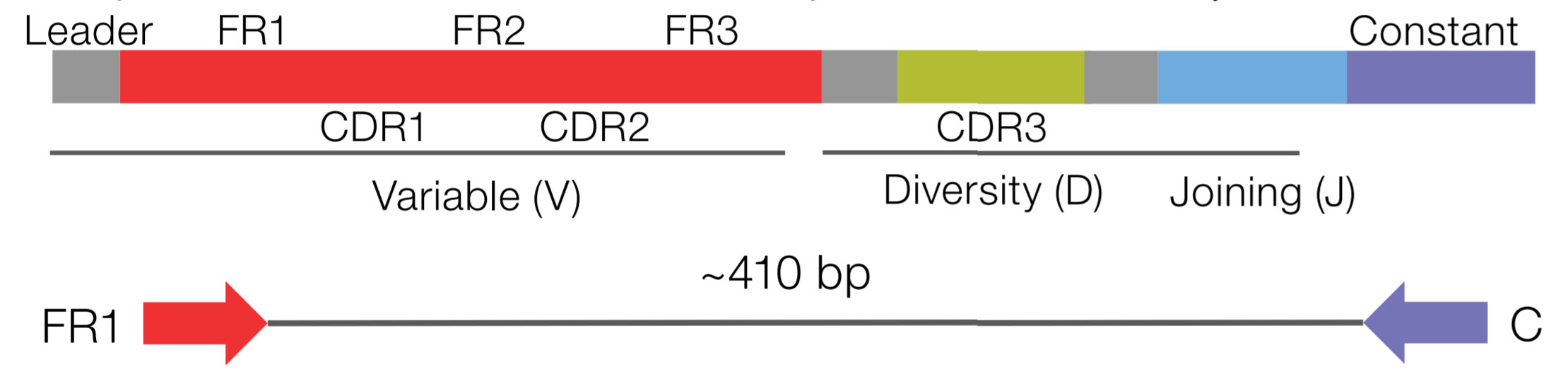
This improved understanding led to the immunoediting hypothesis, an extension of the cancer immunosurveillance hypothesis that includes three phases: elimination of neoplastic cells by the immune system equilibrium, in which the immune system controls but does not eliminate cancer and escape, in which cancer progresses to clinically detectable disease ( 5, 12). In addition, selection pressure from antitumor immune responses contributes to the evolution of immune resistant tumors. While the immune system can recognize and destroy tumor cells, tumors can evade and suppress immune responses. In the decades since, we have acquired a better understanding of the complex interactions and mutual influence between tumor cells and the host immune system ( 10, 11). Thus, it was shown that lymphocytes and IFNγ collaboratively function to suppress tumor formation. Furthermore, mice deficient in both RAG2 and IFNγ receptor developed more spontaneous tumors than mice deficient only in RAG2 ( 9). It was then demonstrated in 2001 that RAG2-deficient (and therefore lymphocyte-deficient) mice developed more tumors than wild-type mice and developed them earlier, and this was true of both spontaneous and chemically induced tumors ( 9). The same study also showed that IFNγ receptor–deficient p53-deficient mice developed spontaneous tumors more rapidly and developed a broader spectrum of them than mice deficient only in p53 ( 8). First, in 1998, it was shown that IFNγ receptor–deficient mice developed more chemically induced tumors than wild-type mice and developed them faster ( 8). The hypothesis was hotly debated in the following decades due to conflicting data and a lack of experimental protocols for directly testing the hypothesis ( 1, 5–7), but eventually, mouse experiments convincingly demonstrated the existence of cancer immunosurveillance and revealed interferon γ and lymphocytes as its key components ( 8, 9). In the mid-1950s, Lewis Thomas and Frank MacFarlane Burnet formalized the cancer immunosurveillance hypothesis, proposing that the immune system could recognize and eliminate tumors via tumor-specific antigens ( 1, 3, 4). Paul Ehrlich first proposed that host defense mechanisms could detect and eliminate neoplastic cells in 1909 ( 1, 2). The review then outlines important questions left unanswered and suggests future directions for the field. This review describes the current state of knowledge based on repertoire sequencing studies conducted on human cancer patients, with a focus on studies of the T-cell receptor beta chain locus. Repertoire sequencing is thus a critical complement to other methods of lymphocyte and immune profiling in cancer. Lymphocyte antigen receptor repertoire deep sequencing data can be used to assess the clonal richness and diversity of lymphocyte populations to track clone members over time, between tissues, and across lymphocyte subsets to detect clonal expansion and to detect the recruitment of new clones into a tissue. Deep sequencing of the repertoire of antigen receptor–encoding genes expressed in a lymphocyte population has become a widely used approach for profiling the population's immune status. A better understanding of lymphocyte population dynamics over the course of disease and in response to therapy is urgently needed to guide therapy decisions and to develop new therapy targets. They are directly targeted by some therapies, and the composition and spatial organization of intratumor T-cell populations is prognostic in some cancer types. Lymphocytes play a critical role in antitumor immune responses.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
